Looking for information about other Dental Topics?
Full Website Index• Animated-Teeth.com •
Best Fluoride Toothpastes for Preventing Cavities
Using a good fluoride toothpaste can significantly reduce your risk of tooth decay.
If you’ve had trouble with getting cavities, you’ll want to make sure that the kind of toothpaste you use is as effective in preventing tooth decay as possible. And in most cases, this means selecting one that’s formulated with fluoride.
Fluoride was the first therapeutic agent (active ingredient) added to toothpaste formulations. And over six decades of clinical research have consistently proven its effectiveness in preventing cavities and reducing decay rates.
Fluoride usage promotes optimal oral health.
Few public health initiatives have had a more profound impact on dental health than the widespread adoption of fluoride. Its introduction has revolutionized oral hygiene, helping individuals worldwide maintain healthier teeth.
Research underscores fluoride’s importance in cavity prevention. For example, the study “Fluoride toothpastes for preventing dental caries in children and adolescents” states that incorporating fluoride toothpaste into a daily brushing routine can reduce cavities by approximately 24% compared to using non-fluoridated products.
states that incorporating fluoride toothpaste into a daily brushing routine can reduce cavities by approximately 24% compared to using non-fluoridated products.
This level of reduction even applies to widely available over-the-counter fluoride toothpastes, like those mentioned on this page. This evidence highlights fluoride’s vital role in protecting teeth from decay and reinforces its place as a cornerstone of modern oral hygiene practices.
How does fluoride prevent tooth decay?
Fluoridated toothpaste provides anticavity protection in two primary ways.
Protection #1 – Fluoride promotes tooth remineralization.
You may not have heard of it, but there’s a process called “remineralization” that plays a critical role in slowing down cavity formation. Think of it as reversing the damage caused by tooth decay.
In this process, minerals that were previously leached from a tooth during demineralization are redeposited onto areas where they were lost. Essentially, remineralization helps “heal” these damaged areas, restoring the tooth’s structure.
(FYI: The constant interplay between demineralization and remineralization is an interesting one. The links above lead to our page that explains this phenomenon in greater detail.)
How fluoride enhances remineralization.
Research shows that fluoride enhances the remineralization process, helping to reduce mineral loss during demineralization. Here’s how it works.
(The details of our summary have been taken from the paper “Demineralization–remineralization dynamics in teeth and bone” and the textbook “Dental Caries: The Disease and Its Clinical Management.”
and the textbook “Dental Caries: The Disease and Its Clinical Management.” )
)
Without fluoride –
When the oral environment’s pH drops below approximately 5.5, tooth demineralization begins. This involves the loss of hydroxyapatite, the mineral that gives tooth enamel its strength.
Once the pH rises back to neutral levels, remineralization starts, redepositing hydroxyapatite onto the tooth.
With fluoride –
In the presence of fluoride, demineralization still occurs as pH drops. However, during remineralization, a new compound called fluorohydroxyapatite forms and is deposited onto the tooth.
Fluorohydroxyapatite is more resistant to acid than hydroxyapatite, and the process of remineralization can begin at a lower pH. This means healing starts sooner, even before the oral environment fully returns to neutral pH conditions.
As a result, the presence of fluoride leads to less overall mineral loss from the tooth, effectively strengthening enamel and protecting against decay.
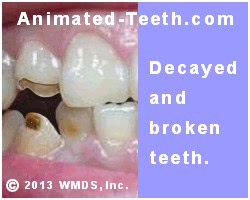
Extensive tooth decay.
Protection #2 – Fluoride makes teeth more decay-resistant.
A new, stronger tooth mineral is created.
When remineralization occurs in the presence of fluoride, a new type of tooth mineral called fluorohydroxyapatite (also referred to as fluoroapatite or fluorhydroxyapatite) is formed. This compound is significantly “harder” and more resistant to decay than the original minerals present in teeth. This unique property is a critical factor in cavity prevention.
Why Is Fluorohydroxyapatite Important?
Most of a tooth’s structure is composed of hydroxyapatite and carbonated hydroxyapatite, which are naturally vulnerable to acid damage. When fluoride is present during remineralization, fluorohydroxyapatite replaces these minerals, offering enhanced protection against acid attacks from bacteria.
Fluorohydroxyapatite is superior because it is more resistant to the acidic environment created by plaque bacteria. While hydroxyapatite begins to dissolve at a pH of approximately 5.5, fluorohydroxyapatite can withstand acidity levels as low as pH 4.5 (values may vary depending on individual oral conditions). This increased resistance significantly reduces the risk of mineral loss and tooth decay.
This enhanced protection ensures that teeth remain stronger and more resilient over time, emphasizing the critical role fluoride plays in maintaining oral health.
Key features to look for in a fluoride toothpaste.
1) The kind of fluoride compound used.
Types of fluoride compounds in toothpaste.
Several types of fluoride compounds can be included in toothpaste formulations as the effective agent (active ingredient) for preventing tooth decay. While around 20 fluoride compounds are used in the European Union, only three are approved for use in the United States, and they cannot be combined. These are:
- Stannous Fluoride (tin difluoride, SnF2)
Stannous fluoride was the first fluoride compound incorporated into commercial toothpaste as an anticavity agent, debuting in 1956.
- Sodium Fluoride (NaF)
- Sodium Monofluorophosphate (SMFP, Na2PO3F)
(FYI: We’ve based this section of our page on information taken from the monograph “An Introduction to Toothpaste – Its Purpose, History and Ingredients.” )
)
Comparing fluoride compounds.
Sodium Monofluorophosphate vs. Stannous Fluoride vs. Sodium Fluoride
No single fluoride compound is inherently better than the others. In terms of cavity protection, all three are equally effective. Each compound serves as a delivery mechanism for fluoride ions (F⁻), which promote remineralization and strengthen enamel when released into the oral environment.
Why manufacturers choose specific fluoride compounds.
The choice of fluoride compound in a toothpaste formulation often depends on the compatibility of other ingredients. Certain components, such as abrasives or other active agents, can interfere with the fluoride compound’s ability to deliver effective fluoride levels. Manufacturers select the fluoride type that best complements the overall formulation to ensure efficacy.
The added advantage of stannous fluoride.
Beyond its role as a source of fluoride ions, stannous fluoride offers an additional benefit: antibacterial properties. This dual action helps toothpaste formulated with stannous fluoride provide both cavity protection and bacterial control, without requiring extra antibacterial agents.
2) The toothpaste’s fluoride concentration.
- By FDA regulation (USA), any dentifrice that contains more than 1500 ppm (parts per million) of fluoride is classified as prescription medicine. Some prescription products have a concentration as high as 5000 ppm F-.
- Over-the-counter (OTC) toothpastes typically contain between 1000 – 1500 ppm F-. The vast majority of which are targeted toward the 1000 – 1100 ppm end of the range.
- Some toothpastes designed for young children may be formulated with a fluoride concentration of 500 ppm. However, the effectiveness of these lower-concentration products is questionable.
(FYI: We’ve based this section of our page on information taken from the paper titled “Dentifrices – an update.” )
)
Which is better, a 1000 or 5000 ppm fluoride toothpaste?
a) 5000 ppm toothpastes (prescription products)
Prescription-strength dentifrices with higher fluoride concentrations offer enhanced cavity protection and are more effective for individuals with high decay rates. However, they are not suitable for everyone. For instance, they are inappropriate for young children with developing teeth or adults with minimal cavity risk.
In the U.S., high-fluoride dentifrices require a dentist’s prescription, so it is up to your dentist to determine if you require one.
b) 1000 ppm toothpastes (OTC products).
Regular over-the-counter (OTC) fluoride toothpastes are highly effective at preventing cavities. A review of dental literature by Walsh concluded that a dentifrice must deliver a concentration of at least 1000 ppm F- to produce significant anticavity benefits.
Most OTC toothpastes meet this standard, with fluoride concentrations typically ranging from 1000 to 1500 ppm (with 1000 ppm being the more common level).
For individuals with a history of cavities who haven’t been using a fluoride toothpaste, starting with an OTC product can reduce their decay rate by approximately 24%. This simple change can be an important first step toward improved oral health.
This simple change can be an important first step toward improved oral health.
How to determine the fluoride concentration in toothpaste.
1) Check the Label: Look for the type of fluoride compound listed on the product and its percentage.
2) Understand Common Equivalents:
a) 1000 ppm F⁻ equivalents (the minimum for adults and older children) –
Stannous fluoride: 0.4% | Sodium fluoride: 0.22% | Sodium monofluorophosphate: 0.76%
b) 1100 ppm F⁻ equivalents (stronger OTC products) –
Stannous fluoride: 0.454% | Sodium fluoride: 0.243% | Sodium monofluorophosphate: 0.836%
Prescription-Strength Formulations –
For high-strength toothpastes, sodium fluoride is typically used. | 1.1% sodium fluoride
Which are the best fluoride toothpastes?
Look for the ADA Seal.
Selecting an effective fluoride toothpaste to reduce cavities is really quite easy. Simply look for products that display the American Dental Association’s (ADA) “Seal of Acceptance” on their packaging. This seal guarantees that the product has undergone rigorous testing and has been proven safe and effective in preventing tooth decay.
The ADA Seal of Acceptance.

Products that display this logo are effective anticavity dentifrices.
Details you should know about ADA-accepted toothpastes.
- All toothpastes awarded the ADA Seal of Acceptance have undergone rigorous testing to confirm they deliver a therapeutic level of fluoride. Manufacturers are required to provide research evidence demonstrating their product’s effectiveness, and each dentifrice must contain at least 90% of the fluoride amount listed on its label.
- All over-the-counter (OTC) toothpastes with the ADA Seal of Acceptance are recognized as effective treatments for reducing cavities.
- As discussed previously on this page, any of the three fluoride compounds approved for use in U.S. toothpaste formulations (stannous fluoride, sodium fluoride, and sodium monofluorophosphate) can be used to create an effective product. No single compound is best in terms of fluoride ion release or cavity prevention.
a) The best over-the-counter (OTC) fluoride toothpastes.
Here is a small sampling of the more than 50 toothpastes that have earned the ADA Seal of Acceptance for cavity protection. We do not consider any single product to be superior but, instead, we view all of these as equally effective.
- Crest Pro-Health
- Colgate Total SF
- Crest Plus Cavity Protection
- Colgate Cavity Protection
- Colgate Kids Maximum Cavity Protection
- Tom’s of Maine Natural Fluoride Toothpaste for Children
- Crest Gum Detoxify
- Crest Gum and Enamel Repair
- Parodontax Daily Fluoride Anticavity and Antigingivitis
- Made By Dentists Fluoride Ultra Whitening Toothpaste
- Tom’s of Maine Simply White
- Sensodyne Toothpaste
Please note, the specific products we included in our list were only chosen to illustrate that high-quality fluoride dentifrices can also be formulated to tackle other tasks too. This includes:
- Plaque/Gingivitis Control
- Enamel Erosion Control
- Oral Malodor Control
- Tooth Sensitivity Control
- Stain Removal (whitening)
- Products for small children.
We should also note that multi-care toothpastes (products designed to provide several of the protections mentioned above) can be an excellent, and perhaps the best, choice. We discuss the best multi-care toothpastes here.
If you’d like to consider even more brands.
If you’d like to explore additional options, the following brands each offer at least one toothpaste product that has earned the ADA Seal of Acceptance.
Aim, AloeSense, Aquafresh, Benco Pro-Sys, Burt’s Bees, Closys, Colgate, Cool Wave, Crest, CVS, DTI, Enamel Rebuild, Euthymol, FireFly, Freshmint, Hello, Made by Dentist, Moon, Oraline, Parodontax, Quip, Sensodyne, SprinJene, Tom’s of Maine,
Search the ADA database for yourself.
If you’d like to explore all of the fluoride toothpaste products that have earned the ADA Seal of Acceptance, you can search their database here: ADA Accepted Fluoride Toothpastes
b) The best prescription fluoride toothpastes.
The ADA Seal program no longer evaluates “professional dental products,” which appears to include prescription fluoride toothpastes containing 5000 ppm F⁻. This assumption is based on the absence of any such products in the current list of accepted items.
If your dentist has a strong preference for a specific dentifrice brand, they will indicate it on your prescription. Otherwise, you can assume they believe that any generic product will be equally effective.
Prescription fluoride toothpastes (5000 ppm F-).
Each of these products is a sodium fluoride (1.1%) dentifrice: PreviDent 5000, SF 5000 Plus, Clinpro 5000, Denta 5000 Plus
Precautions about fluoride toothpaste use in children.
Swallowing fluoridated toothpaste during a child’s tooth-forming years can result in a permanent condition called dental fluorosis. In mild cases, this condition causes tooth discoloration, ranging from opaque white spots to brown stains. In more severe instances, it can also affect the enamel’s surface texture, creating visible irregularities.
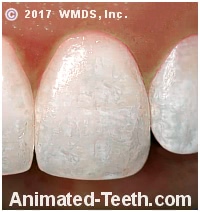
Generalized, severe dental fluorosis.
Parents should be mindful of this risk and take appropriate precautions to minimize the likelihood of fluorosis formation when fluoridated toothpaste (1000 ppm F-) is used:
- Monitor the amount of toothpaste your child uses. A pea-sized amount is generally considered sufficient for children ages 3 and up. For younger children, use just a smear (rice-sized) application.
- Teach and encourage proper brushing habits, including spitting out excess toothpaste rather than swallowing it.
- Based on typical tooth development timelines, the most critical period for the risk of fluorosis occurs from birth to around 6 or 7 years of age. By approximately age 8, a child’s permanent teeth have largely formed, and the risk of developing fluorosis is no longer a concern.
Recommendations for fluoridated toothpaste use by children.
Note: In all cases and at all ages, the above rules need to apply.
For toddlers (under 3 years) –
With supervision, children in this age group may safely use a smear of toothpaste containing up to 1,000 ppm fluoride.
For children aged 3–6 years –
With supervision, children in this age group may safely use a pea-sized application of toothpaste containing up to 1,000 ppm fluoride.
Older children –
The use of pea-sized application of fluoridated toothpaste containing 1,000 to 1,500 ppm fluoride is considered appropriate.
FAQs about fluoridated toothpaste.
What is fluoride and how does it help prevent cavities?
Fluoride is a naturally occurring mineral that’s added to many dental products, like toothpaste, because of its well-documented ability in helping to prevent tooth decay.
Fluoride helps prevent cavities in two primary ways:
- It helps to “strengthen” tooth enamel through a process called remineralization.
- It inhibits the growth and metabolism of the bacteria that cause cavities.
These benefits make fluoride an essential component of oral care products and a cornerstone of modern cavity prevention strategies.
You can find more details about how fluoride works here.
How can I tell if a toothpaste contains fluoride?
An easy way to check is by looking for the ADA Seal of Acceptance on the packaging. All ADA approved toothpastes contain an optimal level of fluoride for preventing cavities.
Another method is to examine the toothpaste’s ingredient list. In the U.S., you should find one of the following fluoride compounds listed: Stannous Fluoride, Sodium Fluoride, or Sodium Monofluorophosphate.
Is there an important difference between stannous fluoride, sodium fluoride, and sodium monofluorophosphate?
No, all three fluoride compounds can be used to create an equally effective anticavity toothpaste. The choice of which compound to use often depends on its compatibility with other ingredients in the toothpaste formulation.
Are prescription-strength fluoride toothpastes more effective than OTC options?
Yes, in the sense that they deliver a higher concentration of fluoride ions, which enhances the cavity-reversing remineralization process. However, not everyone requires the additional level of protection that a high-strength toothpaste provides.
Who should use prescription fluoride toothpaste?
Individuals at higher risk of tooth decay are often candidates for high-strength fluoride toothpaste. Common risk factors include dry mouth, high sugar intake, poor oral hygiene, a history of cavities, extensive dental restorations, advanced age, or undergoing cancer treatment.
Since these products require a prescription, your dentist will determine whether they are suitable for your specific needs.
How much fluoride is in regular over-the-counter toothpaste?
While products vary, OTC toothpastes typically contain fluoride concentrations ranging from 1000 to 1500 ppm. At this level, these products are considered safe and effective for both children and adults when used as directed.
Is fluoride toothpaste safe for children?
Yes, when used as directed, OTC fluoridated toothpastes are safe for children. Two primary concerns are ensuring that the child uses an appropriate amount of toothpaste (not too much) and that they spit rather than swallow while brushing.
For young children, direct supervision is essential to ensure these guidelines are followed. If fluoride toothpaste is ingested during the period of tooth formation, it can lead to fluorosis, a condition that causes tooth staining. Here’s further details about precautions with children.
What is the ADA Seal of Acceptance, and why is it important?
Earning the ADA Seal of Acceptance is a voluntary process in which manufacturers, such as toothpaste makers, submit documentation to the American Dental Association demonstrating that their product is both safe and effective for its intended claims.
The seal signifies that the manufacturer is committed to producing a quality product, and its presence confirms that the product meets high standards for safety and efficacy.
Can fluoride toothpaste prevent existing cavities from getting worse?
Possibly, it simply depends on how advanced the decay is. In the case of “incipient” lesions (also termed “white spot lesions“), the additional fluoride provided by the toothpaste may help to reverse or arrest the decay process.
However, once a cavity has significantly damaged the tooth’s surface, especially if a hole has begun to form, it is too late for toothpaste alone to be effective.
Does fluoride toothpaste also help with gum disease or sensitivity?
Yes, fluoride toothpaste can be beneficial for both purposes. Stannous fluoride, the compound used in some toothpaste formulations, has antibacterial properties that promote gum health. Additionally, regular use of fluoridated toothpaste helps to build a protective coating on root surfaces, which can reduce sensitivity over time.
What’s next?
If you’re looking for more information about toothpaste, we’d like to suggest our page Which are the best multi-care toothpastes? Jump
Or, if you’re looking for other topics about maintaining your oral health, scroll on down a few lines to see our ‘What’s Next?’ menu. Thanks for visiting.
Last reviewed: December 24, 2024
Author: Paul Cotner, DMD — retired dentist.
Published by: WMDS, Inc. — owner of Animated-Teeth.com.
Educational information only — not a substitute for professional dental care.
Page references sources:
Abou Neel EA, et al. “Demineralization–remineralization dynamics in teeth and bone”
ADA. Accepted Over-the-Counter Products.
Davies R, et al. “Dentifrices – an update.”
Davies RM, et al. “The rational use of fluoride toothpaste.”
Fejerskov O, et al. “Dental Caries The Disease and Its Clinical Management. Chapter: Biofilms in caries development.”
Lippert F. “An Introduction to Toothpaste – Its Purpose, History and Ingredients.”
Marinho VCC, et al. Fluoride toothpastes for preventing dental caries in children and adolescents
Walsh T, et al. Fluoride toothpastes of different concentrations for preventing dental caries in children and adolescents.
